What is Insulator – Classification, Properties, Examples and Uses?
Hello friends! Welcome back to ElectroDuino. This blog is based on What is Insulator – Classification, Properties, Examples, and Uses? Here we will discuss, What is an Insulator, Structure, Different States, Classification, Properties, Examples, and Uses.
What is Insulator
Insulators are one type of material or substance, which prevents the flow of current through them. Because these types of materials or substances have no free electrons in their atomic structure, that’s why the electrons are unable to move from atom to atom. In the other words, we can say that the insulator is a poor conductor (as of electricity or heat), it has high resistivity that prevents the flow of current. These types of materials do not allow to transfer of electrons from particle to particle.
Basic structure of Insulator material
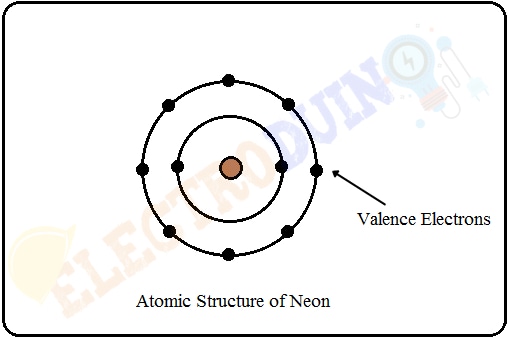
Most of the materials are insulators, which outermost shell of atomic structure has eight or nearer to eight valence electrons. Due to the eight or nearer to eight valence electrons, the atoms of these materials are in a stable condition and they have no free electrons. Also, the forbidden gap between the conduction band and valence band of the insulator is more. So these materials are offered very high resistance. For example, the atomic structure of the gaseous insulating material neon is shown in the below figure.
How it Works
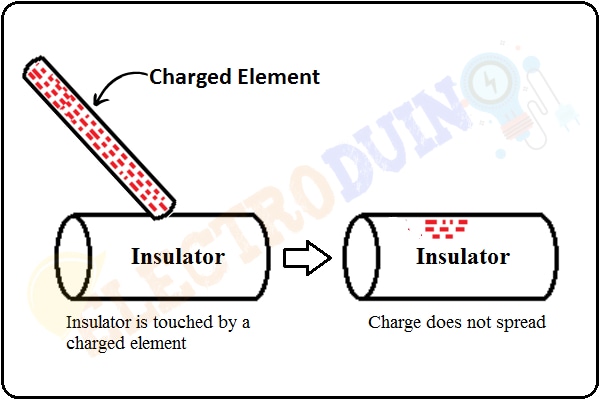
The insulating materials do not permit to transfer of charge across the entire surface of the material. If a charged element is touched to a specific point on insulator material, then this charge does not distribute evenly (spread) across the entire surface of this insulator. This charge will remain at the initial location of the material. This because there is no free electron flow in this material.
For example, glass is a good insulator of heat and electric current, so when we apply heat or electric current to one end of the glass, then we can observe that the heat or electric current does not reach the other end.
Different States of Insulator
The insulator materials are existing in three States, these are Solids, Liquids, and Gaseous. The following table showing the classification of the insulator materials according to Substances and Materials, with examples.
| Different States | Examples of insulator materials |
| Solids | Dry wood, glass, rubber, cotton, silk, Mica, slate, porcelain, rayon, bakelight, paper, and cellulose materials, etc. |
| Liquid | Mineral oil, Linseed oil, refined hydrocarbon mineral oils, spirit, and synthetic varnishes, etc |
| Gaseous | Dry air, carbon dioxide, argon, nitrogen, etc. |
Properties of a Good Insulator
- Resistivity
The resistivity of an insulator should be very high, that’s why electric current does not flow through it. So it can be used for safety equipment of electrical instruments.
- Dielectric Strength:
Its dielectric strength should be very high, so it can be used in the high voltage application.
- Mechanical strength
It must have high mechanical strength so that it does not damage during work. And it will be able to withstand the tension and weight of the conductor.
- Does not affected by nature
Good Insulation material should not be affected by moisture, water, and temperature
- It should be free from impurities.
- Least thermal expansion.
- Its costs should be cheap and easily available
Uses of Insulators
Insulators are mainly used for preventing electric current. So these are highly used in electrical and electronic appliances, equipment, and circuits for safety purposes. These are used in,
- Use to make the coating of cable and wire.
- Use to make the electric switch box
- Use to separate the electric conductors.
- To make the lever of electrical switches
- To make the body of electric appliances

