What is EMF or Electromotive Force? Definition, Symbol, Unit, Formula
Hello friends! Welcome back to ElectroDuino. This blog is based on EMF or Electromotive Force. Here we will discuss what is EMF or Electromotive Force, Definition, Symbol, Unit, Dimension, and Formula.
.What is EMF or Electromotive Force?
Definition: Electromotive force is defined as the maximum electric potential difference developed by any source of electrical energy such as a battery, generator, or photovoltaic cell.
As suggest by the name electromotive force, we think the word “force” means that it is one kind of force, but it is somewhat misleading because EMF is not a force, it is basically Work that transfers energy. In other words, the electromotive force (EMF) is defined as the amount of work done in the energy transformation or conversion from one to another and the amount of electricity that passes through the electrical source, such as the battery or generator.
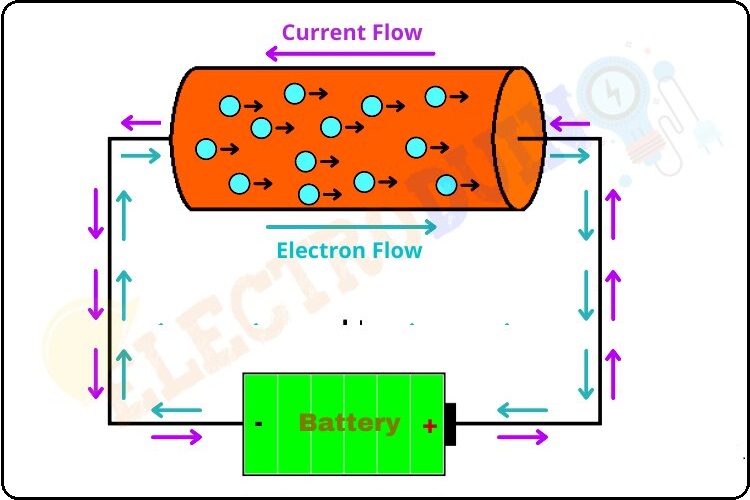
Describe Electromotive Force or EMF
For example, When a battery is connected to a conductor it makes electrons flow from the negative terminal to the positive terminal of the battery through the conductor, which gives some electrical energy to the conductor. The battery is an electrochemical cell, So the work done by the battery converts chemical energy inside it into electrical energy.
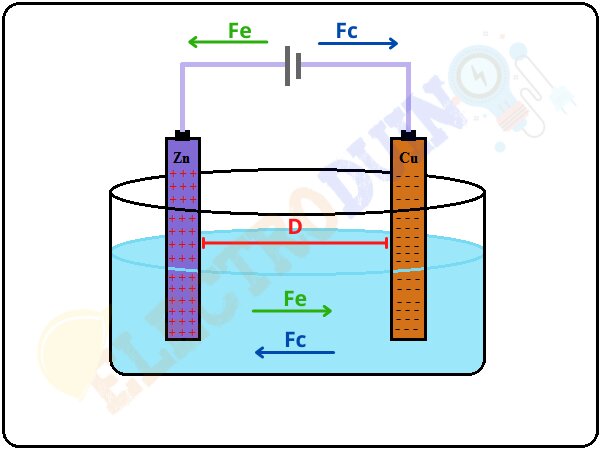
We know that a battery is made of electrolytes and electrodes, whose chemical reaction drives the transfer of positive and negative ions. Let us assume, the force on the ions is Fc and the distance between the two terminals is D, so the work done is equal to the FcD.
Work done = FcD
We also know that this chemical force inside the battery is balanced by an electric force Fe that is equal to Fc.
Fc=Fe
Therefore the work done is also equal to Fed.
Work done = FeD
Now the EMF is defined as work done per unit charge. Therefore in this case EMF will be equal to FeD/q.
EMF = FeD/q
Symbol for Electromotive Force
Electromotive force or EMF is denoted by (E) and it is also denoted by the symbol ε.
Unit of Electromotive Force or EMF
If an electric charge Q passes through a device (device without internal resistance), and gains an energy W. Then net electromotive force or EMF for that device is the energy gained per unit charge. So we can write
EMF = energy gained (W) / unit charge (Q)
= Joules / Coulombs = Volt
So the SI unit of electromotive force is Volt (V), which is equivalent to a joule per coulomb.
Dimension of Electromotive Force
Electromotive Force, EMF (ε) = Work Done / Charge
We know that the dimensional formula of work done = M1 L2 T-2 and for charge = I1 T1
So, the dimensional formula of Electromotive Force = M1 L2 T-2 / I1 T1
= M1 L2 T-3 I-1
Where, M = Mass, I = Current, L = Length, T = Time
Formula for Electromotive Force
The formula for electromotive force or EMF is given by
EMF (ε) = voltage of the cell + (current across the circuit x internal resistance of the cell)
ε = V + Ir
Where,
- V = the voltage of the cell
- I = the current across the circuit
- r = the internal resistance of the cell
- ε = the electromotive force