What is Conductor – Types, Properties, Examples and Uses?
Hello friends! Welcome back to ElectroDuino. This blog is based on What is Conductor – Types, Properties, Examples, and Uses?. Here we will discuss What is a Conductor, Different States, Properties, Examples of Conductors, and their Properties and Uses.
What is Conductor
In electrical and electronic components, Conductors are one type of material or substance, which permits the flow of current through them. Because these types of material or substances have free electrons in the outermost shell of the atoms, these electrons are loosely bound and easily moving from one atom to another. These types of materials are allowed to transfer electrons from particle to particle. Most of the metals are conductors, whose have 1,2, or 3 electrons in the outer shell of the atomic structure.
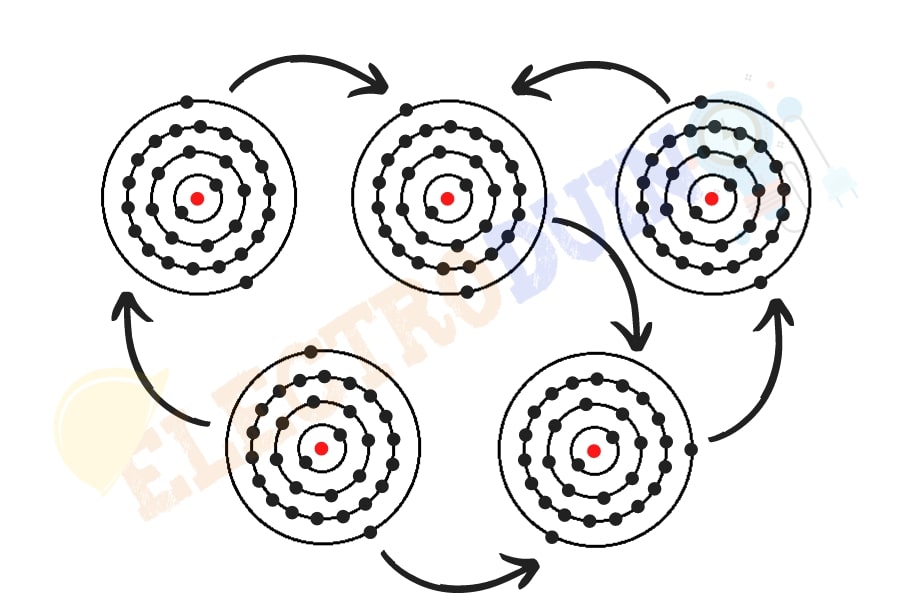
The conducting materials are permits to transfer charge across the entire surface of the material through the free movement of electrons. If a charged conductor is touched to a specific point on another conductor, then this charge quickly spread across the entire surface of this conductor. A charged object will always distribute its charge until the overall repulsive forces between excess electrons are minimized. For example, copper is a good conductor of heat, so when we heating one end of copper results in a quick temperature rise on the other end.
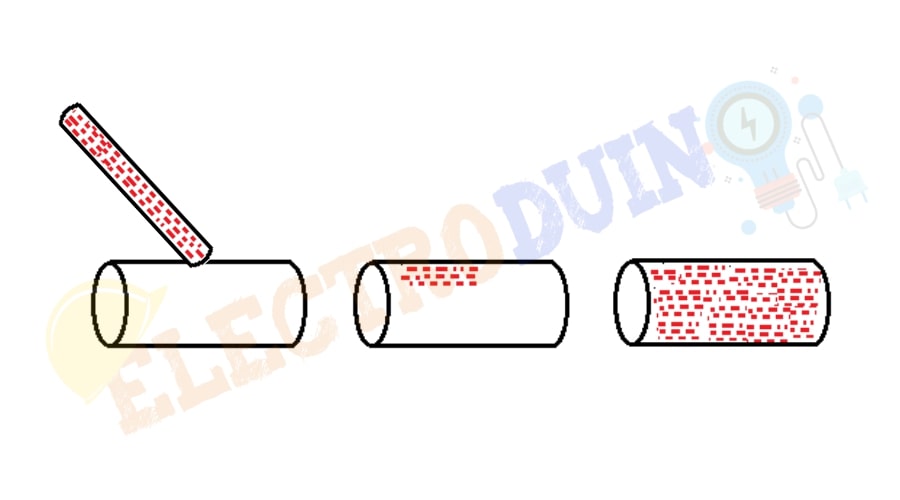
Note: We all know that the electrons are carry a negative electric charge, so these electrons are easily repelled (repulsion) by an applied negative electric charge and attracted by a positive charge.
Different States of Conductive material
The conductor materials are existing in three States, these are Solids, Liquids, and Gaseous.
| Solids | Silver, Copper, Iron, Aluminium, Gold, etc. are examples of Solid conductor materials. Mostly these are used to making wires, cables, and electrical/electronic equipment. |
| Liquid | Mercury (Hg) is a good conductor, which stays Liquid state at room temperature. The solution of Sulphuric acid (H₂SO₄), Hydrochloric acid (HCl) in Water (H2O) are also good conductors. Vinegar contains acetic acid (CH₃COOH), lemon juice contains Citric acid (C₆H₈O₇), and Copper sulfate (CuSO4) solution, these are also conductors. |
| Gaseous | Pure Gas is not a good conductor of electricity. When gases are ionized then they become a good conductor of electricity. For example, Neon gas is used to make neon lights. |
Properties of a Good Conductor
- Resistance of Electric Conductor
The resistance of a good conductor should be as low as possible to avoid the heat effect. If a conductor has high resistance, then it losing energy by produce heat. Conductors conductivity depends on its resistance, if resistance is increase then conductivity will decrease and resistance is decreased then conductivity will increase. The resistance of an ideal conductor should be zero. Practically high resistance and low resistance conductor both are used to making different types of elements. For example, the conductor having low resistivity/high conductivity is used to making transmission lines, electrical contact, and using in the winding of armature, etc. The conductors having high resistivity/low conductivity are used for making filament, heater, Ovens, etc.
- Electric Field inside the Conductor
The electrical field inside a good conductor should be zero. If the electric field exists inside the conductor, it will extract a force on electrons and accelerate the electrons.
- Charge Density inside the Conductor
This electric charge should not exist inside the conductor. The mutual electrostatic repulsion force between electrons, which demands that the electrons must be as far as possible. This repulsion force sends the electrons to the surface of the conductor. For this reason, the electric charge should not exist inside the conductor. So, the charge density inside the conductor should be zero.
- Ductility of Conductor
A good conductor should be ductile. So it can be used to make thin wires, these wires are used in winding of armature and coil.
- A conductor should be tough and strong, which is used to make overhead transmission lines.
- A conductor should be malleable, which helps to convert it into thin sheets.
- A good conductor should not be influenced by external weather conditions, like rain, sunlight, sun temperature, summer, winter, etc.
- It should be cheap, that can be easily purchased to use it.
Examples of Conductors and their Properties and Uses
| Name of Conductors | Description |
Properties | Uses | ||
| Melting Point (°C) | Resistivity (Ω-m) | Temperature Coefficient at 20°C | |||
| Silver |
Silver is a white-colored, malleable, shiny, expensive, and high conductive material. It is the best conducting material of electricity because it has high number of free electrons in its atom. So, its resistivity is very low and its conductivity is very high. |
960 | 1.59x10-8 | 0.0038 | It is more expensive than other materials so it cannot be used for general purposes. It is normally used in relay, satellite, starters, conduct points, etc. |
| Copper | Copper is a brownish-orange colored conductor material having high malleability, ductility, high tensile strength, low specific resistance, inexpensive and high conductivity. But, its conductivity is less than silver. | 1086 | 1.68x10-8 | 0.00428 |
Copper can be easily soldered and due to high tensile strength, it can be converted into wires and cables that are used in power transmission, power distribution, telecommunications, electronics circuitry. Also used in windings of electric motors, transformers, inductors, generators, and headphones, etc. |
| Gold | It is a yellowish-colored, highly conductive, lustrous, and high expensive material. | 1063 | 2.44x10-8 | 0.0034 |
It is highly conductive and anti-corrosive, but due to its high cost it is rarely used in every electronics component and appliance. But a small amount of gold is used in some sophisticated electronic devices like mobile phones, GPS modules, PDA’s, Digital calculators, Smart TVs, etc. |
| Aluminium |
Aluminium is a silvery-white colored lightweight, malleable, ductility, low resistance, and anti-corrosive material. its conductivity less than copper, due to its huge availability and the less costly it is used over copper. But the aluminum forms an electrically resistant oxide layer in the electrical joints surface when exposed to the atmosphere, which can cause the connection to overheat. |
657 | 2.82x10-8 | 0.004013 |
It is a soft metal, so can be used for overhead transmission lines after made alloyed with some other materials like magnesium, silicon, or iron. It also used to make Antenna. |
| Tungsten |
Tungsten is a dark gray conductive material having high resistivity, high melting point, and high strength. It is very hard and it has high thermal stability that’s why it does not burn easily at a high temperature. |
3422 | 5.6x10-8 | 0.0051 |
It is highly used in light industry for making filaments of bulbs and fluorescent tubes. |
| Zinc |
Zinc is a bluish-silver-colored soft, anti-corrosive conductive material. |
420 | 5.9x10-8 | 0.0037 |
It is non-reactive in air or water. For this reason, one of the most important applications of zinc is in the galvanization process, in this process a thin layer of zinc coats on other metals surface such as iron or steel, that protects from rusting. it is also used to make electrodes of primary cells and batteries. |


Pingback: What is Electric Current – its Unit, Symbol, Types, and measurement »